What Is Endometrial Cancer?
Cancer can affect the uterus, the hollow, pear-shaped organ where a baby grows. The uterus is lined with a special tissue called the endometrium. When cancer grows in this lining, it is called endometrial cancer. Most cancers of the uterus are endometrial cancer.
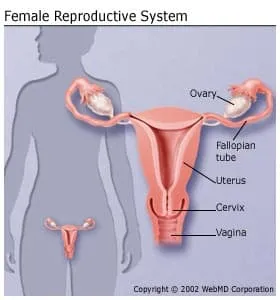
If left untreated, endometrial cancer can spread to the bladder or rectum, or it can spread to the vagina, fallopian tubes, ovaries, and more distant organs. Fortunately, endometrial cancer grows slowly and, with regular checkups, is usually found before spreading very far.
What Does the Type of Endometrial Cancer Mean?
If you get diagnosed with endometrial cancer, your doctor might tell you it’s type 1 or type 2.
About 80% of endometrial cancers are type 1. This kind usually doesn’t form, grow, or spread very quickly.
Experts think it’s caused by having too much of the sex hormone estrogen. Sometimes it develops from a condition called atypical hyperplasia, which is when too many cells grow in the endometrium.
The prognosis for people with type 1 endometrial cancer is usually good. Prognosis is an estimate of how the course of the disease will go for you.
Up to 20% of endometrial cancer is type 2. This type is more likely to grow and spread outside of your uterus.
Experts don’t think that having too much estrogen causes it.
The prognosis (outlook) isn’t as good compared with type 1. Doctors usually need to treat type 2 endometrial cancer with more intense treatments.
Grading Endometrial Cancer
If you get a diagnosis of type 1 or type 2 endometrial cancer, your doctor might talk to you about its “grade.” For most cancers, the grade (also called “differentiation”) is a number that tells you how abnormal your cancer cells look under a microscope. A lower number means the cancer cells are more normal-looking, and in general that means your cancer is more likely to be slow-growing.
For instance, grades 1 and 2 endometrioid adenocarcinoma are considered type 1 endometrial cancer. Your doctor may call these “lower grade.”
Grade 3 endometrioid tumors are considered type 2 endometrial cancer. The doctor might call it higher grade or “poorly differentiated.”
Are There Different Kinds of Endometrial Cancer?
Yes. Doctors divide them up into different categories based on how their cells look under a microscope. The most common type of endometrial cancer is a kind of adenocarcinoma (cancer that forms in glandular tissue) called endometroid cancer.
Endometrioid cancer starts in the glands of your endometrium (uterine lining), and under the microscope it looks very similar to normal uterine lining (endometrium). It varies from person to person in terms of how severe it is.
Some other kinds of endometrial cancer are:
Uterine papillary serous carcinoma: This rarer form of endometrial and uterine cancer starts in the lining of the uterus. It tends to come back even if your doctor spots it early.
Uterine clear cell carcinoma: This form is even more rare, accounting for less than 5% of cases.
Uterine carcinosarcoma: This rare kind of endometrial and uterine cancer also makes up less than 5% of cases, but it tends to be aggressive. That means it forms, grows, or spreads quickly.
What Are the Risk Factors for Endometrial Cancer?
Endometrial cancer usually happens in women past menopause. More than 95% of endometrial cancer happens in women over 40. Postmenopausal women have a high risk for endometrial cancer if they:
- Got their first period early
- Went through menopause late
- Are obese
- Have diabetes or high blood pressure
- Have few or no children
- Have a history of infertility, irregular periods, or abnormal cells in the endometrium (called endometrial hyperplasia)
- Have a family history of endometrial, colorectal, or breast cancer
Women taking the drug tamoxifen to treat or prevent breast cancer have a slightly higher risk of endometrial cancer. But women who have taken birth control pills are only half as likely to have endometrial cancer after menopause.
Women who take estrogen-only hormone replacement therapy have a higher risk of developing endometrial cancer. So women who have not had a hysterectomy should not be taking estrogen-only hormone replacement therapy.
Rare ovarian tumors can make estrogen and increase a woman’s chance of having endometrial cancer.
High-fat diets, especially containing red meat, can increase the risk of cancer, including endometrial and colon cancer.
What Are the Survival Rates for Endometrial Cancer?
It depends on how far your cancer has spread when you’re first diagnosed with it. To show the survival rates for endometrial cancer, the American Cancer Society uses a database that groups this type of cancer into three stages that describe how far it has spread:
Localized means there’s no sign that your endometrial cancer has spread beyond your uterus.
Regional means the disease has spread to nearby areas or lymph nodes.
Distant means the cancer has spread far away, to parts of the body like your lungs, liver, or bones.
If you have localized endometrial cancer, the 5-year relative survival rate is 96%. That means that people who have your type of cancer are about 96% as likely as people who don’t have that cancer to live for at least 5 years, on average, after they’re diagnosed.
The 5-year relative survival rate for regional endometrial cancer is 71%. For distant endometrial cancer it’s 20%.
Keep in mind that other things affect your outlook, like your age, general health, and how well you respond to treatment.
And if you’ve just gotten diagnosed with endometrial cancer, you may have a better outlook than these numbers suggest. That’s because treatments get better over time, and these numbers are based on women who got diagnosed and treated at least 5 years earlier.
Can Endometrial Cancer Be Prevented?
Most endometrial cancer cannot be prevented. But there are certain things women can do to lower their risk. Taking birth control lowers the risk, but first talk with a doctor about possible pros and cons. Being healthy, eating well, and watching your weight may help lower the risk.

