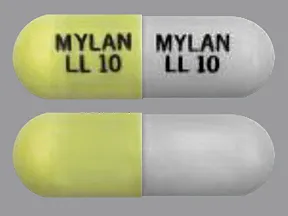
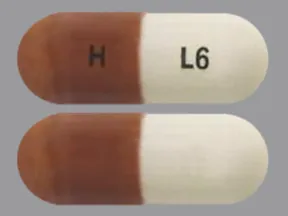
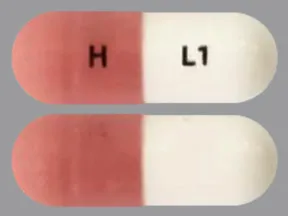
See also Warning section.
Diarrhea, stomach/abdominal pain, nausea/vomiting, loss of appetite, constipation, dizziness, dry skin, dry mouth, or trouble sleeping may occur. If any of these effects last or get worse, tell your doctor or pharmacist promptly.
People using this medication may have serious side effects. However, you have been prescribed this drug because your doctor has judged that the benefit to you is greater than the risk of side effects. Careful monitoring by your doctor may decrease your risk.
Tell your doctor right away if you have any serious side effects, including: mental/mood changes (such as depression), increased thirst/urination, shaking (tremor), numbness/tingling of arms/legs, fast/slow/irregular heartbeat, symptoms of heart failure (such as shortness of breath, swelling ankles/feet, unusual tiredness, unusual/sudden weight gain).
Some people treated with this medication may rarely get other cancers. Consult your doctor for more details.
Lenalidomide sometimes causes side effects due to the rapid destruction of cancer cells (tumor lysis syndrome). To lower your risk, your doctor may add a medication and tell you to drink plenty of fluids. Tell your doctor right away if you have symptoms such as: low back/side pain (flank pain), signs of kidney problems (such as painful urination, pink/bloody urine, change in the amount of urine), muscle spasms/weakness.
People taking lenalidomide may have worsening of their symptoms (tumor flare reaction). If you have tender/swollen lymph nodes, fever, pain, or rash, contact your doctor right away.
This drug helps many people live longer. However, some people who have used lenalidomide have died sooner than expected, especially if they had high white blood cell or tumor cell counts. Talk with your doctor about the risks and benefits of using this medication.
Lenalidomide may rarely cause serious (possibly fatal) liver disease. Get medical help right away if you have any symptoms of liver damage, such as: nausea/vomiting that doesn't stop, loss of appetite, severe stomach/abdominal pain, yellowing eyes/skin, dark urine.
Lenalidomide can commonly cause a rash that is usually not serious. However, you may not be able to tell it apart from a rare rash that could be a sign of a severe reaction. Get medical help right away if you develop any rash.
A very serious allergic reaction to this drug is rare. However, get medical help right away if you notice any symptoms of a serious allergic reaction, including: rash, itching/swelling (especially of the face/tongue/throat), severe dizziness, trouble breathing.
This is not a complete list of possible side effects. If you notice other effects not listed above, contact your doctor or pharmacist.
In the US - Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088 or at www.fda.gov/medwatch.
In Canada - Call your doctor for medical advice about side effects. You may report side effects to Health Canada at 1-866-234-2345.