What Is Endometriosis?
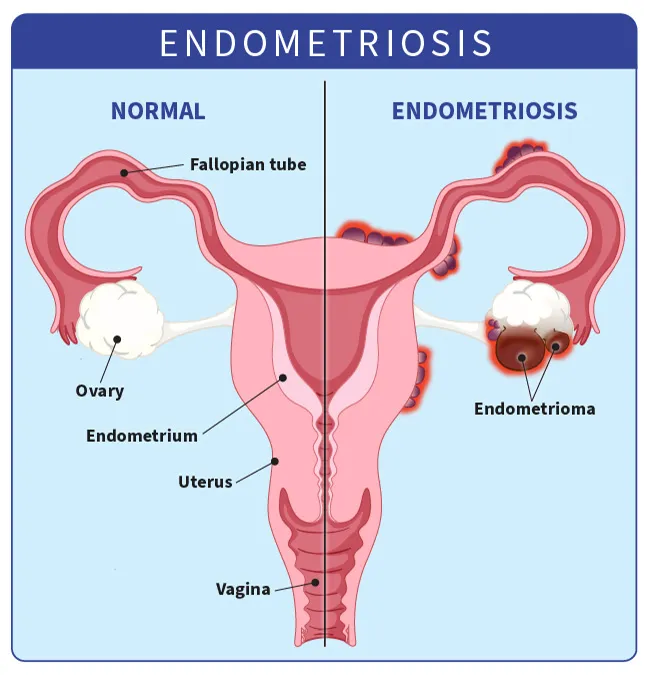
Endometriosis happens when tissue similar to the tissue that lines your uterus grows outside of the uterus.
This tissue acts as regular uterine tissue does during your period — it breaks apart and bleeds at the end of the cycle. But this blood has nowhere to go. Surrounding areas may become inflamed or swollen. You might have scar tissue and lesions.
Endometriosis is most common in your ovaries.
Types of Endometriosis
Based on what area of the pelvis or abdomen it affects, endometriosis can be grouped into four main types:
Superficial peritoneal endometriosis. The peritoneum is a thin membrane that lines your abdomen and pelvis. It also covers most of the organs in these cavities. In this type, the endometrial tissue attaches to the peritoneum. This is the least severe form.
Endometriomas. Also called chocolate cysts, these are dark, fluid-filled cysts. They vary in size and can appear in different parts of your pelvis or abdomen, but they’re most common in the ovaries.
Deeply infiltrating endometriosis (DIE). In this type, the endometrial tissue invades the organs either within or outside your pelvic cavity. This can include your ovaries, rectum, bladder, and bowels. It’s rare, but sometimes a lot of scar tissue can bond organs so they become stuck in place. This condition is called a frozen pelvis. But this only happens to 1%-5% of people with endometriosis.
Abdominal wall endometriosis. In some cases, endometrial tissue can grow on the abdominal wall. The cells may attach to a surgical incision, like one from a C-section.
Endometriosis Symptoms
You might not notice any signs of endometriosis. When you have them, they can include:
- Back pain during your period
- Severe menstrual cramps
- Pain when pooping or peeing, especially during your period
- Unusual or heavy bleeding during periods
- Blood in your stool or urine
- Diarrhea or constipation
- Painful sex
- Fatigue that won’t go away
- Trouble getting pregnant
Your symptoms don’t necessarily reflect the severity of your endometriosis. You can have a lot of extra tissue with no symptoms, or a small amount that causes a lot of symptoms.
Endometriosis Causes
Doctors don’t know exactly what causes endometriosis. Some experts think menstrual blood that contains endometrial cells may pass back through your fallopian tubes and into your pelvic cavity, where the cells stick to your organs. This is called retrograde menstruation.
Your genes could also play a role. If your mom or sister has endometriosis, you’re more likely to get it. Research shows that it tends to get worse from one generation to the next.
Some people with endometriosis also have immune system disorders. But doctors aren’t sure whether there’s a link.
Other reasons may include:
Transformation of peritoneal cells. Experts think that hormones or immune factors promote the transformation of peritoneal cells — cells that line the inner side of your abdomen — into endometrial-like cells.
Embryonic cell transformation. Hormones such as estrogen may transform embryonic cells — cells in the earliest stages of development — into endometrial-like cell implants during puberty.
Surgical scar implantation. After a surgery, such as a hysterectomy or C-section, endometrial cells may attach to a surgical incision.
Endometrial cell transport. The blood vessels or tissue fluid (lymphatic system) may transport endometrial cells to other parts of the body.
Risk Factors for Endometriosis
Some things can raise your risk for endometriosis. You’re more likely to get it if you:
- Have a close relative (for example, your parent, sibling, or aunt) with endometriosis
- Have a low BMI
- Have short menstrual cycles (a gap of less than 28 days between the start of one period and the next)
- Have heavy periods that last more than a week
- Were very young when you got your first period
- Went through menopause at an older age
- Have never given birth
- Have high estrogen levels
- Have a health condition that stops blood from flowing normally during your period
Endometriosis Complications
About half of people with endometriosis have a hard time getting pregnant. To get pregnant, your ovary must release an egg into the fallopian tube, where a sperm can fertilize it. But endometriosis tissue can block the tube. When this happens, sperm can’t reach the egg, and you can’t get pregnant. It’s also possible that endometriosis tissue could harm the sperm or egg.
Despite these challenges, many people with mild or moderate endometriosis can still get pregnant and have a baby. But you might need the help of fertility treatment, especially as you get older.
Depending on where the extra tissue grows, endometriosis can cause problems with other organs. It could impact:
- Your bowel and bladder: You might notice blood in your poop or pee.
- Your kidneys: This could cause swelling.
- Your diaphragm or lungs: This could lead to difficulty breathing or chest pain.
Severe endometriosis pain can affect your quality of life. Some people struggle with anxiety or depression. Medical treatments and mental health care can help.
Endometriosis may raise your risk of ovarian cancer or another cancer called endometriosis-associated adenocarcinoma. But the odds of getting cancer from endometriosis are extremely low.
Endometriosis and Fertility
Endometriosis is the leading cause of infertility. It affects about 5 million people in the U.S., many in their 30s and 40s. Nearly 2 of every 5 people who can’t get pregnant have it.
If endometriosis interferes with your reproductive organs, your ability to get pregnant can become an issue:
- When endometrial tissue wraps around your ovaries, it can block your eggs from releasing.
- The tissue can block sperm from making its way up your fallopian tubes.
- It can stop a fertilized egg from sliding down your tubes to your uterus.
A surgeon can fix those problems, but endometriosis can make it hard for you to conceive in other ways:
- It can change your body’s hormonal chemistry.
- It can cause your body’s immune system to attack the embryo.
- It can affect the layer of tissue lining your uterus where the egg implants itself.
Your doctor can surgically remove the endometrial tissue. This clears the way for the sperm to fertilize the egg.
If surgery isn’t an option, you might consider intrauterine insemination (IUI), which involves putting your partner’s sperm directly into your uterus.
Your doctor may suggest pairing IUI with “controlled ovarian hyperstimulation,” which means using medicine to help your ovaries put out more eggs. People who use this technique are more likely to conceive than those who don’t.
In vitro fertilization (IVF) is another option. It can raise your chances of conceiving, but the statistics on IVF pregnancies vary.
Endometriosis Diagnosis
Your doctor might suspect endometriosis based on your symptoms. To confirm it, they can do the following tests:
Pelvic exam. Your doctor might be able to feel cysts or scars behind your uterus.
Imaging tests. An ultrasound, a CT scan, or an MRI can make detailed pictures of your organs.
Laparoscopy. Your doctor makes a small cut in your belly and inserts a thin tube with a camera on one end (called a laparoscope). They can see where and how big the lesions are. This is usually the only way to confirm if you have endometriosis.
Biopsy. Your doctor takes a sample of tissue, often during a laparoscopy, and a specialist looks at it under a microscope to confirm the diagnosis.
Endometriosis Stages
There are different ways to measure endometriosis. The most widely used scale is from the American Society of Reproductive Medicine. Doctors assign points according to the spread of the endometrial tissue, its depth, and the areas of your body that are affected.
Based on the results, the condition can be divided into four stages:
Stage I or minimal. There are a few small implants, wounds, or lesions. They may be found on your organs or the tissue lining your pelvis or abdomen. There’s little to no scar tissue.
Stage II or mild. There are more implants than in stage 1. They’re also deeper in the tissue, and there may be some scar tissue.
Stage III or moderate. There are many deep implants. You may also have small cysts on one or both ovaries and thick bands of scar tissue called adhesions.
Stage IV or severe. This is the most widespread. You have many deep implants and thick adhesions. There are also large cysts on one or both ovaries.
Experts don’t know why some people have more severe cases than others. Endometriosis stages don’t take pain or symptoms into account. And it doesn’t always go from one stage to the next. If left untreated, it can either remain the same or get worse or better over time.
Questions for Your Doctor
If you’ve been diagnosed with endometriosis, you might want to ask questions such as:
- Why is endometriosis painful?
- What can I do to control my endometriosis symptoms?
- Do I need medication? How does it work?
- What are the side effects of medication for endometriosis?
- Will endometriosis affect my sex life?
- How do birth control pills affect endometriosis?
- If I'm having trouble getting pregnant, could fertility treatments help? What about surgery?
- Can surgery stop my symptoms?
- What might happen if I do nothing? Can endometriosis go away without drugs or surgery?
- Will it last my whole life?
- Should I consider joining a clinical trial?
- How often do I need to see a doctor?
Endometriosis Treatments
There’s no cure for endometriosis. Treatments usually include surgery or medication. You might need to try different treatments to find what helps you feel better.
Pain medicine. Your doctor may recommend an over-the-counter pain reliever. Nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen (Advil, Motrin) or naproxen (Aleve) work for many people. If these don’t ease your pain, ask about other options.
Hormones. Hormonal therapy lowers the amount of estrogen your body makes and can stop your period. This helps lesions bleed less so you don’t have as much inflammation, scarring, and cyst formation. Common hormones include:
- Birth control pills, patches, and vaginal rings
- Gonadotropin-releasing hormone (Gn-RH) agonists and antagonists, such as elagolix sodium (Orilissa) or leuprolide (Lupron)
- Progestin-only contraceptives
- Danazol (Danocrine)
Surgery. Your doctor might recommend surgery to take out as much of the affected tissue as possible. In some cases, surgery helps ease symptoms and can make you more likely to get pregnant. Your doctor might use a laparoscope or do a standard surgery that uses larger cuts. Sometimes, pain comes back after surgery.
In the most severe cases, you may need a surgery called a hysterectomy to take out your ovaries, uterus, and cervix. But without them, you can’t get pregnant later.
Home Remedies for Endometriosis
Warm baths, hot water bottles, and heating pads can give quick relief from endometriosis pain. Over time, lifestyle changes like these might also help:
Eat right. Research has shown a link between endometriosis and diets that are low in fruits and vegetables and high in red meat. Some experts think the high amount of fat in meat such as beef encourages your body to produce chemicals called prostaglandins, which may lead to more estrogen production. This extra estrogen could cause excess endometrial tissue to grow.
Add more fresh fruits and vegetables by making them the heart of your meals. Stocking your refrigerator with prewashed and precut fruits and vegetables can help you eat more of both.
Research has also found foods rich in omega-3 fatty acids, such as salmon and walnuts, may be helpful. One study suggested that people who ate the highest amount of omega-3 fatty acids were 22% less likely to develop endometriosis than those who ate the least amount.
By comparison, those who ate the most trans fats had a 48% higher risk than those who ate the least. So, the type of fat you eat may matter, but more research is needed.
Also, avoid alcohol and caffeine. Drinking caffeinated coffee and soda seems to increase your odds of developing endometriosis, although researchers aren’t sure why. Alcohol is also associated with a higher risk.
Exercise regularly. There are a lot of reasons exercise is a great way to manage your endometriosis. Working out encourages your heart to pump blood to all your organs, improving circulation and helping nutrients and oxygen flow to all your systems.
People who exercise may also have less estrogen and lighter periods, which can help improve their symptoms of endometriosis over time. What's more, studies have shown that the more time you devote to high-intensity exercises such as running or biking, the less likely you are to ever get endometriosis.
Exercise helps reduce stress. And because it releases brain chemicals called endorphins, it can actually relieve pain. Even just a few minutes of physical activity that makes you breathe hard or sweat can create that effect.
Lower-intensity workouts such as yoga can be beneficial, too, as they help stretch the tissues and muscles in your pelvis for pain relief and stress reduction.
Manage stress. Researchers think stress can make endometriosis worse. In fact, the condition itself might cause stress because of the severe pain and other side effects.
Finding ways to manage stress -- whether it’s through yoga or meditation, or by simply carving out time for self-care -- can help ease symptoms. It may also be helpful to see a therapist who can offer tips for dealing with stress.
Look at alternative therapies. Although there isn’t enough research that supports the use of alternative natural therapies for endometriosis, some people find relief from their symptoms through these techniques, including:
- Acupuncture
- Herbal medicine
- Ayurveda
- Chiropractic care
- Massage
If you’re interested in trying an alternative therapy, be sure to talk to your doctor first, especially if you’re considering taking over-the-counter supplements. They could have side effects that you don’t know about. And never exceed the recommended dosage or take more than one supplement at a time.
Takeaways
Endometriosis is a condition where uterine-like tissue grows outside the uterus. It can cause painful cramps, heavy bleeding, fatigue, and other period-related symptoms. You also might have trouble getting pregnant. Treatment can help you manage these symptoms and, if you want, get pregnant.
Endometriosis FAQs
Is endometriosis curable?
There’s no way to cure endometriosis, but over-the-counter pain medicines and hormone therapy can help manage it. Some people with severe endometriosis get surgery to remove the extra tissue, ovaries, or uterus. But sometimes, endometriosis continues after surgery.
Can you live with endometriosis without treatment?
You don’t have to get treatment for endometriosis if you don’t want it. Over-the-counter NSAIDs such as ibuprofen and naproxen sodium can help with cramps. So can warm baths and heating pads. But untreated endometriosis can lead to worsening pain and trouble getting pregnant.
Is endometriosis a lifelong condition?
Sometimes, endometriosis gets better on its own. Symptoms tend to improve after menopause.
How can you test for endometriosis?
If your doctor thinks you might have endometriosis, they’ll give you a physical exam and ask a few questions. They might put a gloved finger in your vagina to feel if you have any cysts or other abnormalities. They also might suggest an ultrasound, MRI, or laparoscopy.
