Deep brain stimulation (DBS) is a treatment for people with conditions like multiple sclerosis (MS), Parkinson's disease, and essential tremor. It’s a way to “turn off” parts of your brain without destroying them.
During the procedure, a doctor places the tip of an electrode in the thalamus (for tremor and multiple sclerosis) or in the globus pallidus or subthalamic nucleus (for Parkinson's disease). The electrode stays in your brain. A wire connects it to a pacemaker-like device put under the skin of your chest. The device generates electrical shocks.
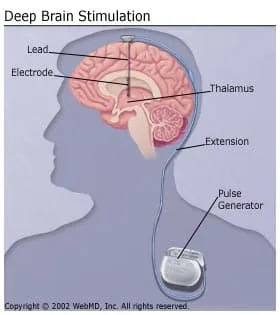
DBS replaces older surgeries that destroy parts of your brain, either the thalamus (thalamotomy) or the globus pallidus (pallidotomy). You can still get these surgeries, but the risks are high. If the surgeon is off by even a fraction of an inch, the procedure may not work and you could have severe complications like paralysis or vision and speech loss.
What Are the Advantages of DBS?
Deep brain stimulation offers many benefits over surgery. The electrical stimulation is adjustable, while surgical destruction isn’t. The electrode has four metal contacts that can be used in many different combinations. Even if one electrode contact isn’t in the exact location, one of the others or some combination of them will be close to the proper target. As your response changes over time, doctors can adjust the stimulation without doing more surgery.
There isn’t much chance you’ll benefit from future surgeries, but your doctor can turn the deep brain stimulator off if they want to try other therapies.
How Does DBS Help With Multiple Sclerosis?
The main use is to control a severe tremor related to the disease. Deep brain stimulation won’t help other MS-related problems like vision loss, sensations, or weakness.
Can Deep Brain Stimulation Cure Multiple Sclerosis?
No. Electrical stimulation doesn’t cure MS or prevent it from getting worse. It eases tremor related to MS.
Is Deep Brain Stimulation Considered Experimental?
No. The FDA has approved DBS to treat Parkinson's disease and essential tremor. It’s also approved for treating dystonia, a type of movement disorder marked by abnormal postures and twisting motions.
The FDA hasn’t specifically approved deep brain stimulation of the thalamus to treat multiple sclerosis. But this doesn’t mean it’s experimental or your insurance won’t cover it. Many treatments used every day aren’t FDA-approved for a specific medical condition.
Who Should Consider Deep Brain Stimulation?
Before you schedule surgery, give medication a try. If drugs control your symptoms, you don’t need DBS. If you aren’t sure whether DBS is right for you, talk to a movement disorders expert or a neurologist who has experience with movement disorders.
Where Do You Get Deep Brain Stimulation?
Doctors perform deep brain stimulation in a medical center where there’s a team of experts to care for you. This means neurologists and neurosurgeons with experience and specialized training in these types of treatments. Always ask a doctor how many times they’ve performed a specific procedure.

