Uses
This medication is used alone or with other medications to prevent nausea and vomiting caused by cancer drug treatment (chemotherapy) and radiation therapy. It is also used to prevent and treat nausea and vomiting after surgery. Ondansetron works by blocking one of the body's natural substances (serotonin) that causes vomiting.
How to use ondansetron oral
This medication is dissolved on top of the tongue. It is not meant to be chewed or swallowed like other tablet forms.
Dry your hands before using this medication. This medication may come in a bottle or a blister pack. If using the blister pack, peel back the foil on the blister pack to remove a tablet. Do not push the tablet through the foil. Immediately after removing the tablet, place it on the tongue. Allow it to dissolve completely, then swallow it with saliva. You do not need to take this product with water. Doing so may increase your chance of getting a headache.
To prevent nausea from chemotherapy, take this medication usually within 30 minutes before treatment begins. To prevent nausea from radiation treatment, take this medication 1 to 2 hours before the start of your treatment. To prevent nausea after surgery, take ondansetron 1 hour before the start of surgery. This medication may be taken with or without food. However, your doctor may tell you not to eat before chemotherapy, radiation, or surgery.
Take any other doses as directed by your doctor. Ondansetron may be taken up to 3 times a day for 1 to 2 days after your chemotherapy or radiation treatment is finished. If you are taking this medication on a prescribed schedule, take it regularly in order to get the most benefit from it. To help you remember, take it at the same times each day.
The dosage is based on your medical condition and response to treatment. The dosage for children may also be based on age and weight. Take this medication exactly as directed. Do not take more medication or take it more often than prescribed. Ask your doctor or pharmacist if you have questions.
Tell your doctor if your condition does not get better or if it gets worse.
Side Effects
Headache, lightheadedness, dizziness, drowsiness, tiredness, or constipation may occur. If these effects last or get worse, tell your doctor promptly.
To reduce the risk of dizziness and lightheadedness, get up slowly when rising from a sitting or lying position.
Remember that this medication has been prescribed because your doctor has judged that the benefit to you is greater than the risk of side effects. Many people using this medication do not have serious side effects.
Tell your doctor right away if you have any serious side effects, including: stomach pain, muscle spasm/stiffness, vision changes (such as temporary loss of vision, blurred vision).
Get medical help right away if you have any very serious side effects, including: chest pain, slow/fast/irregular heartbeat, severe dizziness, fainting.
This medication may increase serotonin and rarely cause a very serious condition called serotonin syndrome/toxicity. The risk increases if you are also taking other drugs that increase serotonin, so tell your doctor or pharmacist of all the drugs you take (see Drug Interactions section). Get medical help right away if you develop some of the following symptoms: fast heartbeat, hallucinations, loss of coordination, severe dizziness, severe nausea/vomiting/diarrhea, twitching muscles, unexplained fever, unusual agitation/restlessness.
A very serious allergic reaction to this drug is rare. However, get medical help right away if you notice any symptoms of a serious allergic reaction, including: rash, itching/swelling (especially of the face/tongue/throat), severe dizziness, trouble breathing.
This is not a complete list of possible side effects. If you notice other effects not listed above, contact your doctor or pharmacist.
In the US -
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088 or at www.fda.gov/medwatch.
In Canada - Call your doctor for medical advice about side effects. You may report side effects to Health Canada at 1-866-234-2345.
Precautions
Before taking ondansetron, tell your doctor or pharmacist if you are allergic to it; or to other anti-nausea serotonin blockers (such as granisetron); or if you have any other allergies. This product may contain inactive ingredients, which can cause allergic reactions or other problems. Talk to your pharmacist for more details.
Before using this medication, tell your doctor or pharmacist your medical history, especially of: irregular heartbeat, liver disease, stomach/intestinal problems (such as recent abdominal surgery, ileus, swelling).
Ondansetron may cause a condition that affects the heart rhythm (QT prolongation). QT prolongation can rarely cause serious (rarely fatal) fast/irregular heartbeat and other symptoms (such as severe dizziness, fainting) that need medical attention right away.
The risk of QT prolongation may be increased if you have certain medical conditions or are taking other drugs that may cause QT prolongation. Before using ondansetron, tell your doctor or pharmacist of all the drugs you take and if you have any of the following conditions: certain heart problems (heart failure, slow heartbeat, QT prolongation in the EKG), family history of certain heart problems (QT prolongation in the EKG, sudden cardiac death).
Low levels of potassium or magnesium in the blood may also increase your risk of QT prolongation. This risk may increase if you use certain drugs (such as diuretics/"water pills") or if you have conditions such as severe sweating, diarrhea, or vomiting. Talk to your doctor about using ondansetron safely.
This drug may make you dizzy or drowsy. Alcohol or marijuana (cannabis) can make you more dizzy or drowsy. Do not drive, use machinery, or do anything that needs alertness until you can do it safely. Avoid alcoholic beverages. Talk to your doctor if you are using marijuana (cannabis).
This medicine may contain aspartame. If you have phenylketonuria (PKU) or any other condition that requires you to restrict your intake of aspartame (or phenylalanine), consult your doctor or pharmacist regarding the safe use of this medicine.
Older adults may be more sensitive to the side effects of this drug, especially QT prolongation (see above).
During pregnancy, this medication should be used only when clearly needed. It may harm an unborn baby. Discuss the risks and benefits with your doctor.
It is unknown if this drug passes into breast milk. Consult your doctor before breastfeeding.
Interactions
Drug interactions may change how your medications work or increase your risk for serious side effects. This document does not contain all possible drug interactions. Keep a list of all the products you use (including prescription/nonprescription drugs and herbal products) and share it with your doctor and pharmacist. Do not start, stop, or change the dosage of any medicines without your doctor's approval.
Some products that may interact with this drug are: apomorphine, tramadol.
The risk of serotonin syndrome/toxicity increases if you are also taking other drugs that increase serotonin. Examples include street drugs such as MDMA/"ecstasy," St. John's wort, certain antidepressants (including SSRIs such as fluoxetine/paroxetine, SNRIs such as duloxetine/venlafaxine), among others. The risk of serotonin syndrome/toxicity may be more likely when you start or increase the dose of these drugs.
Overdose
If someone has overdosed and has serious symptoms such as passing out or trouble breathing, call 911. Otherwise, call a poison control center right away. US residents can call 1-800-222-1222. Canada residents can call 1-844-764-7669.
Do not share this product with others.
Lab and/or medical tests (such as EKG) should be done while you are taking this medication. Keep all medical and lab appointments. Consult your doctor for more details.
Try to take each dose at the scheduled time. If you miss a dose, take it as soon as you remember unless it is near the time for the next dose. In that case, skip the missed dose. Take your next dose at the regular time. Do not double the dose to catch up.
Store in the refrigerator or at room temperature away from light and moisture. Do not store in the bathroom. Keep all medications away from children and pets.
Do not flush medications down the toilet or pour them into a drain unless instructed to do so. Properly discard this product when it is expired or no longer needed. Consult your pharmacist or local waste disposal company.
Images
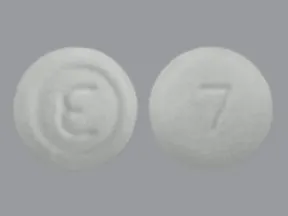
ondansetron 8 mg disintegrating tablet
Color: whiteShape: roundImprint: logo 7This medicine is a white, round, guarana strawberry, tablet imprinted with "logo" and "7".
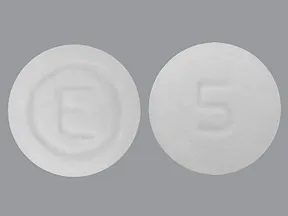
ondansetron 4 mg disintegrating tablet
Color: whiteShape: roundImprint: logo 5This medicine is a white, round, guarana strawberry, tablet imprinted with "logo" and "7".

ondansetron 8 mg disintegrating tablet
Color: whiteShape: ovalImprint: 241This medicine is a white, round, guarana strawberry, tablet imprinted with "logo" and "7".
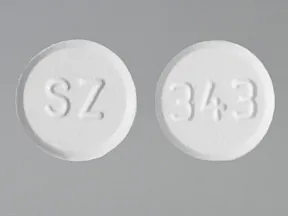
ondansetron 8 mg disintegrating tablet
Color: whiteShape: roundImprint: SZ 343This medicine is a white, round, guarana strawberry, tablet imprinted with "logo" and "7".
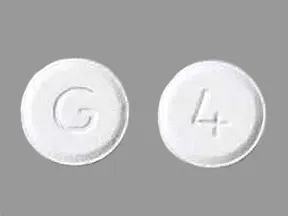
ondansetron 4 mg disintegrating tablet
Color: whiteShape: roundImprint: G 4This medicine is a white, round, guarana strawberry, tablet imprinted with "logo" and "7".
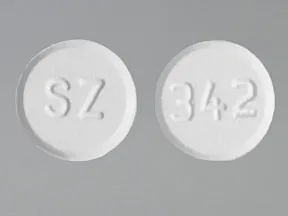
ondansetron 4 mg disintegrating tablet
Color: whiteShape: roundImprint: SZ 342This medicine is a white, round, guarana strawberry, tablet imprinted with "logo" and "7".
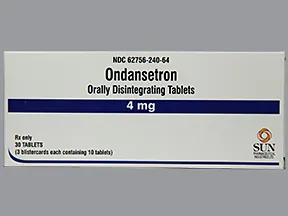
ondansetron 4 mg disintegrating tablet
Color: whiteShape: ovalImprint: 240This medicine is a white, round, guarana strawberry, tablet imprinted with "logo" and "7".

ondansetron 8 mg disintegrating tablet
Color: whiteShape: roundImprint: G 8This medicine is a white, round, guarana strawberry, tablet imprinted with "logo" and "7".
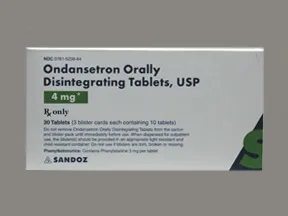
ondansetron 4 mg disintegrating tablet
Color: whiteShape: roundImprint: SZ 342This medicine is a white, round, guarana strawberry, tablet imprinted with "logo" and "7".
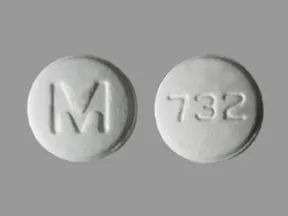
ondansetron 4 mg disintegrating tablet
Color: whiteShape: roundImprint: M 732This medicine is a white, round, guarana strawberry, tablet imprinted with "logo" and "7".
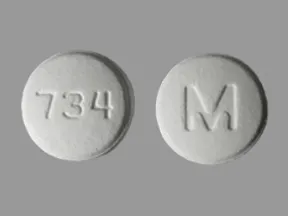
ondansetron 8 mg disintegrating tablet
Color: whiteShape: roundImprint: M 734This medicine is a white, round, guarana strawberry, tablet imprinted with "logo" and "7".
You Might Also Like
Are you currently using ondansetron oral?
This survey is being conducted by the WebMD marketing sciences department.
Selected from data included with permission and copyrighted by First Databank, Inc. This copyrighted material has been downloaded from a licensed data provider and is not for distribution, except as may be authorized by the applicable terms of use.
CONDITIONS OF USE: The information in this database is intended to supplement, not substitute for, the expertise and judgment of healthcare professionals. The information is not intended to cover all possible uses, directions, precautions, drug interactions or adverse effects, nor should it be construed to indicate that use of a particular drug is safe, appropriate or effective for you or anyone else. A healthcare professional should be consulted before taking any drug, changing any diet or commencing or discontinuing any course of treatment.