Uses
See also Warning section.This medication contains hydromorphone in a long-acting form. It is used to help relieve severe ongoing pain (such as due to cancer). Hydromorphone belongs to a class of drugs known as opioid analgesics. It works in the brain to change how your body feels and responds to pain.Do not use the extended-release form of hydromorphone to relieve pain that is mild or that will go away in a few days. This medication is not for occasional ("as needed") use.
How to use Hydromorphone ER
See also Warning section.
Read the Medication Guide and, if available, the Patient Information Leaflet provided by your pharmacist before you start taking this product and each time you get a refill. If you have any questions , ask your doctor or pharmacist.
Take this medication by mouth as directed by your doctor, usually once daily at the same time each day, not as needed for sudden (breakthrough) pain. You may take this drug with or without food. If you have nausea, it may help to take this drug with food. Ask your doctor or pharmacist about other ways to decrease nausea (such as lying down for 1 to 2 hours with as little head movement as possible).
Swallow the tablet whole. Do not crush, break, chew, or dissolve the tablet. Doing so can release all of the drug at once, increasing the risk of side effects.
Hydromorphone comes in a rapid-acting form and a slow-release form. Check with your pharmacist to make sure you are taking the correct form.
The dosage is based on your medical condition and response to treatment. Do not increase your dose, take the medication more often, or take it for a longer time than prescribed. Properly stop the medication when so directed. If you do not take this medication for 3 days in a row, call your doctor before restarting the medication. Your restarting dose may need to be lower.
Before you start using this medication, ask your doctor or pharmacist if you should stop or change how you use your other opioid medication(s). Other pain relievers (such as acetaminophen, ibuprofen) may also be prescribed. Ask your doctor or pharmacist about using hydromorphone safely with other drugs.
Suddenly stopping this medication may cause withdrawal, especially if you have used it for a long time or in high doses. To prevent withdrawal, your doctor may lower your dose slowly. Tell your doctor or pharmacist right away if you have any withdrawal symptoms such as restlessness, mental/mood changes (including anxiety, trouble sleeping, thoughts of suicide), watering eyes, runny nose, nausea, diarrhea, sweating, muscle aches, or sudden changes in behavior.
When this medication is used for a long time, it may not work as well. Talk with your doctor if this medication stops working well.
Though it helps many people, this medication may sometimes cause addiction. This risk may be higher if you have a substance use disorder (such as overuse of or addiction to drugs/alcohol). Take this medication exactly as prescribed to lower the risk of addiction. Ask your doctor or pharmacist for more details.
Tell your doctor if your pain does not get better or if it gets worse, or if you have any new pain.
Side Effects
See also Warning section.
Nausea, vomiting, constipation, lightheadedness, dizziness, or drowsiness may occur. Some of these side effects may decrease after you have been using this medication for a while. If any of these effects last or get worse, tell your doctor or pharmacist promptly.
To prevent constipation, eat dietary fiber, drink enough water, and exercise. You may also need to take a laxative. Ask your pharmacist which type of laxative is right for you.
To reduce the risk of dizziness and lightheadedness, get up slowly when rising from a sitting or lying position.
You may notice an empty tablet shell in your stool. This is harmless because your body has already absorbed the medicine.
Remember that this medication has been prescribed because your doctor has judged that the benefit to you is greater than the risk of side effects. Many people using this medication do not have serious side effects.
Tell your doctor right away if you have any serious side effects, including: interrupted breathing during sleep (sleep apnea), mental/mood changes (such as agitation, hallucinations, confusion), severe stomach/abdominal pain, difficulty urinating, signs of your adrenal glands not working well (such as loss of appetite, unusual tiredness, weight loss).
Get medical help right away if you have any very serious side effects, including: fainting, slow/shallow breathing, severe drowsiness/difficulty waking up, seizures.
A very serious allergic reaction to this drug is rare. However, get medical help right away if you notice any symptoms of a serious allergic reaction, including: rash, itching/swelling (especially of the face/tongue/throat), severe dizziness, trouble breathing.
This is not a complete list of possible side effects. If you notice other effects not listed above, contact your doctor or pharmacist.
In the US -
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088 or at www.fda.gov/medwatch.
In Canada - Call your doctor for medical advice about side effects. You may report side effects to Health Canada at 1-866-234-2345.
Warnings
Hydromorphone has a risk for abuse and addiction, which can lead to overdose and death. Hydromorphone may also cause severe, possibly fatal, breathing problems. To lower your risk, your doctor should have you take the smallest dose of hydromorphone that works, and take it for the shortest possible time. See also How to Use section for more information about addiction.
Ask your doctor or pharmacist if you should have naloxone available to treat opioid overdose. Teach your family or household members about the signs of an opioid overdose and how to treat it.
The risk for severe breathing problems is higher when you start this medication and after a dose increase, or if you take the wrong dose/strength. Taking this medication with alcohol or other drugs that can cause drowsiness or breathing problems may cause very serious side effects, including death. Be sure you know how to take hydromorphone and what other drugs you should avoid taking with it. See also Drug Interactions section. Get medical help right away if any of these very serious side effects occur: slow/shallow breathing, unusual lightheadedness, severe drowsiness/dizziness, difficulty waking up.
Keep this medicine in a safe place to prevent theft, misuse, or abuse. If someone accidentally swallows this drug, get medical help right away.
Before using this medication, women of childbearing age should talk with their doctor(s) about the risks and benefits. Tell your doctor if you are pregnant or if you plan to become pregnant. During pregnancy, this medication should be used only when clearly needed. It may slightly increase the risk of birth defects if used during the first two months of pregnancy. Also, using it for a long time or in high doses near the expected delivery date may harm the unborn baby. To lessen the risk, take the smallest effective dose for the shortest possible time. Babies born to mothers who use this drug for a long time may develop severe (possibly fatal) withdrawal symptoms. Tell the doctor right away if you notice any symptoms in your newborn baby such as crying that doesn't stop, slow/shallow breathing, irritability, shaking, vomiting, diarrhea, poor feeding, or difficulty gaining weight.
Hydromorphone extended-release should be used only if you have been regularly taking moderate to large amounts of opioid pain medication. This medication may cause overdose (even death) if taken by a person who has not been regularly taking opioids.
This product is designed to slowly release hydromorphone. Do not break, crush, dissolve, or chew this medication. Taking broken, crushed, dissolved, or chewed forms of extended-release hydromorphone could cause a fatal overdose.
Precautions
Before taking hydromorphone, tell your doctor or pharmacist if you are allergic to hydromorphone; or to other opioid pain medications (such as hydrocodone, morphine); or if you have any other allergies. This product may contain inactive ingredients (such as sulfites), which can cause allergic reactions or other problems. Talk to your pharmacist for more details.
Before using this medication, tell your doctor or pharmacist your medical history, especially of: brain disorders (such as head injury, tumor, seizures), breathing problems (such as asthma, sleep apnea, chronic obstructive pulmonary disease-COPD), kidney disease, liver disease, mental/mood disorders (such as confusion, depression), personal or family history of a substance use disorder (such as overuse of or addiction to drugs/alcohol), stomach/intestinal problems (such as blockage, stomach or bypass surgery, constipation, diarrhea due to infection, paralytic ileus), difficulty swallowing, difficulty urinating (such as due to enlarged prostate), disease of the pancreas (pancreatitis), gallbladder disease.
This drug may make you dizzy or drowsy. Alcohol or marijuana (cannabis) can make you more dizzy or drowsy. Do not drive, use machinery, or do anything that needs alertness until you can do it safely. Avoid alcoholic beverages. Talk to your doctor if you are using marijuana (cannabis).
Before having surgery, tell your doctor or dentist about all the products you use (including prescription drugs, nonprescription drugs, and herbal products).
Older adults may be more sensitive to the side effects of this drug, especially confusion, dizziness, drowsiness, and slow/shallow breathing.
During pregnancy, this medication should be used only when clearly needed. It may harm an unborn baby. Discuss the risks and benefits with your doctor. (See also Warning section.)
This drug passes into breast milk and may have undesirable effects on a nursing infant. Tell the doctor right away if your baby develops unusual sleepiness, difficulty feeding, or trouble breathing. Consult your doctor before breastfeeding.
Interactions
See also Warning section.
Drug interactions may change how your medications work or increase your risk for serious side effects. This document does not contain all possible drug interactions. Keep a list of all the products you use (including prescription/nonprescription drugs and herbal products) and share it with your doctor and pharmacist. Do not start, stop, or change the dosage of any medicines without your doctor's approval.
Some products that may interact with this drug include: certain pain medications (mixed opioid agonist-antagonists such as butorphanol, nalbuphine, pentazocine), naltrexone, samidorphan.
The risk of serious side effects (such as slow/shallow breathing, severe drowsiness/dizziness) may be increased if this medication is taken with other products that may also cause drowsiness or breathing problems. Tell your doctor or pharmacist if you are taking other products such as other opioid pain or cough relievers (such as codeine, hydrocodone), alcohol, marijuana (cannabis), drugs for sleep or anxiety (such as alprazolam, lorazepam, zolpidem), muscle relaxants (such as carisoprodol, cyclobenzaprine), or antihistamines (such as cetirizine, diphenhydramine).
Check the labels on all your medicines (such as allergy or cough-and-cold products) because they may contain ingredients that cause drowsiness. Ask your pharmacist about using those products safely.
This medication may interfere with certain lab tests (such as amylase/lipase levels), possibly causing false test results. Make sure lab personnel and all your doctors know you use this drug.
Overdose
If someone has overdosed and has serious symptoms such as passing out or trouble breathing, give them naloxone if available, then call 911. If the person is awake and has no symptoms, call a poison control center right away. US residents can call 1-800-222-1222. Canada residents can call 1-844-764-7669. Symptoms of overdose may include: slow/shallow breathing, slow heartbeat, coma.
Do not share this medication with others. Sharing it is against the law.
This medication has been prescribed for your current condition only. Do not use it later for another condition unless told to do so by your doctor. A different medication may be necessary in that case.
If you miss a dose, skip the missed dose. Take your next dose at the regular time. Do not double the dose to catch up. Do not take 2 doses in 24 hours.
Store at room temperature away from light and moisture. Do not store in the bathroom. Keep all medications away from children and pets. See also Warning section.
Do not flush medications down the toilet or pour them into a drain unless instructed to do so. Properly discard this product when it is expired or no longer needed. For more details, read the Medication Guide, or consult your pharmacist or local waste disposal company.
Images

hydromorphone ER 16 mg tablet,extended release 24 hr
Color: light beigeShape: roundImprint: 268This medicine is a light beige, round, film-coated, tablet imprinted with "268".

hydromorphone ER 12 mg tablet,extended release 24 hr
Color: light yellowShape: roundImprint: 267This medicine is a light beige, round, film-coated, tablet imprinted with "268".
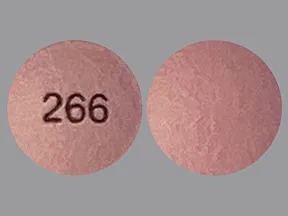
hydromorphone ER 8 mg tablet,extended release 24 hr
Color: light pinkShape: roundImprint: 266This medicine is a light beige, round, film-coated, tablet imprinted with "268".
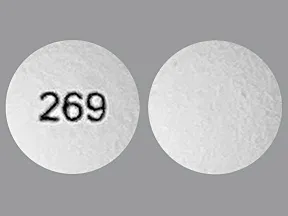
hydromorphone ER 32 mg tablet,extended release 24 hr
Color: whiteShape: roundImprint: 269This medicine is a light beige, round, film-coated, tablet imprinted with "268".
You Might Also Like
Are you currently using Hydromorphone ER?
This survey is being conducted by the WebMD marketing sciences department.
Selected from data included with permission and copyrighted by First Databank, Inc. This copyrighted material has been downloaded from a licensed data provider and is not for distribution, except as may be authorized by the applicable terms of use.
CONDITIONS OF USE: The information in this database is intended to supplement, not substitute for, the expertise and judgment of healthcare professionals. The information is not intended to cover all possible uses, directions, precautions, drug interactions or adverse effects, nor should it be construed to indicate that use of a particular drug is safe, appropriate or effective for you or anyone else. A healthcare professional should be consulted before taking any drug, changing any diet or commencing or discontinuing any course of treatment.