Uses
Diltiazem is used to treat high blood pressure (hypertension) and prevent chest pain (angina). Lowering high blood pressure helps prevent strokes, heart attacks, and kidney problems. When used regularly, diltiazem can decrease the number and severity of episodes of chest pain from angina. It may help increase your ability to exercise.Diltiazem is called a calcium channel blocker. It works by relaxing blood vessels in the body and heart so blood can flow more easily. Diltiazem also lowers your heart rate. These effects help the heart work less hard and lower blood pressure.
How to use Taztia XT
Take this medication by mouth with or without food as directed by your doctor, usually once daily. Swallow the capsules whole. Do not crush or chew the capsules. Doing so can release all of the drug at once and may increase your risk of side effects.
If you have trouble swallowing the capsule, you may open the capsule and carefully sprinkle its contents on a spoonful of soft, cool applesauce just before you take it. Swallow all of the drug/food mixture right away. Do not chew the mixture. Then rinse your mouth and swallow the rinse liquid to make sure that you have swallowed all of the medicine. Do not prepare a supply in advance.
Your doctor may gradually increase your dose. Follow your doctor's instructions carefully. The dosage is based on your medical condition, response to treatment, and other medications you may be taking. Be sure to tell your doctor and pharmacist about all the products you use (including prescription drugs, nonprescription drugs, and herbal products).
Use this medication regularly to get the most benefit from it. To help you remember, use it at the same time each day. Keep taking this medication even if you feel well. Most people with high blood pressure do not feel sick. For the treatment of high blood pressure, it may take 2 to 4 weeks before you get the full benefit of this drug.
This medication must be taken regularly to prevent angina. It should not be used to treat angina when it occurs. Use other medications (such as nitroglycerin placed under the tongue) to relieve an angina attack as directed by your doctor. Consult your doctor or pharmacist for details.
Tell your doctor if your condition does not get better or if it gets worse (for example, your blood pressure readings remain high or increase, your chest pain worsens).
Side Effects
Dizziness, lightheadedness, weakness, nausea, flushing, constipation, and headache may occur. If any of these effects last or get worse, tell your doctor or pharmacist promptly.
To reduce the risk of dizziness and lightheadedness, get up slowly when rising from a sitting or lying position.
Remember that this medication has been prescribed because your doctor has judged that the benefit to you is greater than the risk of side effects. Many people using this medication do not have serious side effects.
Tell your doctor right away if you have any serious side effects, including: nausea/vomiting that doesn't stop, fainting, new or worsening symptoms of heart failure (such as shortness of breath, swelling ankles/feet, unusual tiredness, unusual/sudden weight gain), slow/irregular/pounding/fast heartbeat, mental/mood changes (such as depression, agitation), unusual dreams, severe stomach/abdominal pain, dark urine, yellowing eyes/skin.
A very serious allergic reaction to this drug is rare. However, get medical help right away if you notice any symptoms of a serious allergic reaction, including: rash, itching/swelling (especially of the face/tongue/throat), severe dizziness, trouble breathing.
This is not a complete list of possible side effects. If you notice other effects not listed above, contact your doctor or pharmacist.
In the US -
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088 or at www.fda.gov/medwatch.
In Canada - Call your doctor for medical advice about side effects. You may report side effects to Health Canada at 1-866-234-2345.
Precautions
Before taking diltiazem, tell your doctor or pharmacist if you are allergic to it; or if you have any other allergies. This product may contain inactive ingredients, which can cause allergic reactions or other problems. Talk to your pharmacist for more details.
Before using this medication, tell your doctor or pharmacist your medical history, especially of: certain types of heart rhythm problems (such as sick sinus syndrome/atrioventricular block), liver disease, kidney disease, heart failure.
This drug may make you dizzy. Alcohol or marijuana (cannabis) can make you more dizzy. Do not drive, use machinery, or do anything that needs alertness until you can do it safely. Limit alcoholic beverages. Talk to your doctor if you are using marijuana (cannabis).
Before having surgery, tell your doctor or dentist about all the products you use (including prescription drugs, nonprescription drugs, and herbal products).
Older adults may be more sensitive to the side effects of this drug, especially dizziness, constipation, or swelling ankles/feet.
During pregnancy, this medication should be used only when clearly needed. Discuss the risks and benefits with your doctor.
This drug passes into breast milk and may have undesirable effects on a nursing infant. Consult your doctor before breastfeeding.
Interactions
Drug interactions may change how your medications work or increase your risk for serious side effects. This document does not contain all possible drug interactions. Keep a list of all the products you use (including prescription/nonprescription drugs and herbal products) and share it with your doctor and pharmacist. Do not start, stop, or change the dosage of any medicines without your doctor's approval.
Some products that may interact with this drug include: digoxin, fingolimod, labetalol.
Other medications can affect the removal of diltiazem from your body, which may affect how this medication works. Examples include azole antifungals (such as ketoconazole), cimetidine, macrolide antibiotics (such as erythromycin), rifamycins (such as rifabutin, rifampin), St. John's wort, among others.
This medication can slow down the removal of other medications from your body, which may affect how they work. Examples of affected drugs include aprepitant/fosaprepitant, asunaprevir, buspirone, colchicine, elacestrant, flibanserin, ivabradine, lomitapide, certain benzodiazepines (triazolam, midazolam), among others.
Some products have ingredients that could raise your heart rate or blood pressure. Tell your pharmacist what products you are using, and ask how to use them safely (especially cough-and-cold products, diet aids, or NSAIDs such as ibuprofen/naproxen).
Overdose
If someone has overdosed and has serious symptoms such as passing out or trouble breathing, call 911. Otherwise, call a poison control center right away. US residents can call 1-800-222-1222. Canada residents can call 1-844-764-7669.
Do not share this medication with others. Talk with your doctor about making changes to your lifestyle that may help this medication work better (such as stress reduction programs, exercise, and dietary changes).
Lab and/or medical tests (such as kidney/liver function, pulse, blood pressure, EKG) may be done while you are taking this medication. Keep all medical and lab appointments. Consult your doctor for more details.
There are different brands and types of this medication available. Many do not have the same effects. Do not change brands or types without consulting your doctor or pharmacist.
Have your blood pressure and pulse (heart rate) checked regularly while taking this medication. Learn how to check your own blood pressure and pulse at home, and share the results with your doctor.
If you miss a dose, take it as soon as you remember. If it is near the time of the next dose, skip the missed dose. Take your next dose at the regular time. Do not double the dose to catch up.
Store at room temperature away from light and moisture. Do not store in the bathroom. Keep all medications away from children and pets.
Do not flush medications down the toilet or pour them into a drain unless instructed to do so. Properly discard this product when it is expired or no longer needed. Consult your pharmacist or local waste disposal company.
Images
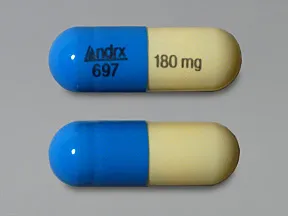
Taztia XT 180 mg capsule,extended release
Color: light blue,buffShape: oblongImprint: Andrx 697 180 mgThis medicine is a light blue buff, oblong, capsule imprinted with "Andrx 697" and "180 mg".

Taztia XT 120 mg capsule,extended release
Color: pinkShape: oblongImprint: Andrx 696 120 mgThis medicine is a light blue buff, oblong, capsule imprinted with "Andrx 697" and "180 mg".

Taztia XT 360 mg capsule,extended release
Color: light blueShape: oblongImprint: Andrx 700 360 mgThis medicine is a light blue buff, oblong, capsule imprinted with "Andrx 697" and "180 mg".

Taztia XT 300 mg capsule,extended release
Color: pink,buffShape: oblongImprint: Andrx 699 300 mgThis medicine is a light blue buff, oblong, capsule imprinted with "Andrx 697" and "180 mg".

Taztia XT 240 mg capsule,extended release
Color: pink,light blueShape: oblongImprint: Andrx 698 240 mgThis medicine is a light blue buff, oblong, capsule imprinted with "Andrx 697" and "180 mg".
Are you currently using Taztia XT?
This survey is being conducted by the WebMD marketing sciences department.
Selected from data included with permission and copyrighted by First Databank, Inc. This copyrighted material has been downloaded from a licensed data provider and is not for distribution, except as may be authorized by the applicable terms of use.
CONDITIONS OF USE: The information in this database is intended to supplement, not substitute for, the expertise and judgment of healthcare professionals. The information is not intended to cover all possible uses, directions, precautions, drug interactions or adverse effects, nor should it be construed to indicate that use of a particular drug is safe, appropriate or effective for you or anyone else. A healthcare professional should be consulted before taking any drug, changing any diet or commencing or discontinuing any course of treatment.